After US and Taiwan, Russia allows Eyesiu’s patent claims of novel formulations of PEA and CsA, likely the best compound to reduce mortality in COVID-19
Pieter J. Gaillard
After US and Taiwan, Russia allows Eyesiu's patent claims on novel formulations of palmitoylethanolamide (PEA) and Cyclosporine A (CsA), which is likely the best compound known to date to reduce mortality in COVID-19
Leiden, The Netherlands, August 23rd, 2020 - Eyesiu Medicines BV, a Leiden Bio Science Park company that creates innovative therapeutics and nutraceuticals based on advanced delivery technology, received “Decision to Grant” letter from the Federal Service for Intellectual Property (ROSPATENT) of Russia for its proprietary formulation and medical use patent application “Pegylated lipid nanoparticle with bioactive lipophilic compound” (2018108203/04(012689), No. 2737893)).
Following USPTO (US15/751,170), and Taiwan (TW201705973A; 105125583), Eyesiu also received the “Decision to Grant” letter from the Federal Service for Intellectual Property (ROSPATENT) of Russia for its patent application ““Pegylated lipid nanoparticle with bioactive lipophilic compound”, while the application is pending in the EU, China, South Korea, Japan and India. The granted claims for the solvent-free and serum-stable nanoparticles comprise two or more phospholipids and at least one of an acylethanolamide (such as the endocannabinoids 2-arachidonoylglycerol (2-AG), arachidonoylethanolamide (AEA or anandamide), oleoylethanolamide (OAE), or palmitoylethanolamide (PEA)) and a macrolide (such as tacrolimus, sirolimus or cyclosporine A), wherein the nanoparticle has a surface comprising a water soluble polymer (such as PEG).
“Being rewarded with an issued patent by Russia, Taiwan and the US for the core of our invention is very gratifying and a validation of Eyesiu’s series of related therapeutics and nutraceutical products in development and available on the market”, comments Dr. Pieter J. Gaillard, founder and CEO of Eyesiu. “This third allowance in a row of Eyesiu’s patent application, now by ROSPATENT, with broad and commercially relevant claims deepens our belief that the technology will also obtain similar product protection in the other geographies where the application is currently pending”, Gaillard adds.
Use of CsA in COVID-19
Of special note is that at the start of the first wave of COVID-19 in Europe, Gaillard already wrote a series of blogs in which he explained how in the immediate term and by using this innovative solvent-free and serum-stable nanotechnology, the existing medicines such as CsA for patients, and lipid-based formulations of food supplements such as PEA for consumers, can already help to combat against COVID-19, the latter by providing targeted support for a healthy diet and lifestyle. See these blogs here
(in Dutch about PEA), and here
(in English about CsA and PEA), and here
(about the PEA hypothesis). Meanwhile, on 15 October 2020, a first study was published in a Lancet article
in which CsA in more than 600 patients with Corona showed that administration of this drug reduces the risk of death by 81%. These promising data were generated by a team at a Spanish hospital in Madrid, who analysed the records of 607 severe COVID-19 patients admitted in March and April during the first wave, when they were treated with one or more potential helpful drugs in critical cases, including CsA, glucocorticoids, tocilizumab, HIV antivirals, and hydroxychloroquine. In this study, CsA was the only drug that clearly reduced deaths
(36 (14%) of the 253 patients who received CsA died, whereas in a closely matched comparison group 105 (almost 30%) of 354 patients who did not get the drug died). Such encouraging outcomes definitely warrant further investigations on the use of CsA in severe COVID-19 patients, while CsA's tolerability may be further improved by Eyesiu's nanotechnology and readily scaled up to widespread use.
Eyesiu’s therapeutics and nutraceuticals
Eyesiu has developed therapeutics, in pre-clinical stage, targeting a wide range of indications in neurology, ophthalmology, dermatology and pulmonology. The company has also developed nutraceuticals (food supplements) that support its users to maintain pain-free and healthier brain, eyes, lungs, skin and/or hair: the first one is a spray bottle with PEA, comprising a lipid-based sublingual, skin and mouth spray formulation for convenient, effective, fast and sustained delivery, and is for sale through Eyesiu’s health care division (Eyesiu Care) via www.PEArlboost.com
About Eyesiu Medicines BV and Eyesiu Care
For more information see www.eyesiu-medicines.com
and www.eyesiu-care.com
For further information, please contact
Eyesiu Medicines BV | Pieter J. Gaillard | info@eyesiu.com

Leiden, The Netherlands, September 3rd, 2024 - Eyesiu Medicines BV, a Leiden Bio Science Park company that creates innovative therapeutics and nutraceuticals based on advanced delivery technology, received the news today that on August 26th 2024 the Korean Intellectual Property Office has granted Eyesiu a patent entitled “Pegylated lipid nanoparticle with bioactive lipophilic compound” ( No. KR20187006989 ). South Korea is the sixth country in a row - following USA, Taiwan, Russia, Japan and India - to provide a granted patent to Eyesiu for its inventions described in the patent application ““Pegylated lipid nanoparticle with bioactive lipophilic compound”. Now the application is only still pending in the EU and China. The Korean Intellectual Property Office granted claims for the solvent-free and serum-stable lipid (nano)particles comprising two neutral phospholipids (DSPE and HSPC) and at least one of palmitoylethanolamide (PEA), an endocannabinoid from the group of acylethanolamides, and a macrolide (such as CsA, tacrolimus or sirolimus), wherein the lipid particle has a surface comprising a water soluble polymer (PEG). Eyesiu’s therapeutics and nutraceuticals Eyesiu has developed therapeutics, in pre-clinical stage, targeting a wide range of indications in neurology, ophthalmology, dermatology and pulmonology. The company has also developed nutraceuticals (food supplements) that support its users to maintain pain-free and healthier brain, eyes, lungs, skin and/or hair: the first one is a spray bottle with PEA, comprising a lipid-based sublingual, skin and mouth spray formulation for convenient, effective, fast and sustained delivery, and is for sale through Eyesiu’s health care division (Eyesiu Care) via www.PEArlboost.com About Eyesiu Medicines BV See www.eyesiu.com for more information, and to see how you can join and own shares in the company, or close a product partnership . For further information, please contact Eyesiu Medicines BV | Pieter J. Gaillard | info@eyesiu.com

Ons lichaam is in staat zichzelf te herstellen. Hippocrates (460–377 v.Chr.) noemde dit ‘physis’, het natuurlijk aanwezige zelfgenezend vermogen. In de meeste gevallen kan je zelfstandig goed genezen van allerlei interne en externe prikkels die ons lichaam constant onder vuur nemen. Zo zijn we langdurig in staat om effectief de brandjes te blussen die veroorzaakt kunnen worden door de meest uiteenlopende trauma’s, infecties, allergenen, straling, chemicaliën, mutaties, metabolieten, ziektes, en ook door de (over)actieve ontstekingsfactoren uit ons eigen afweersysteem. Een gezonde voeding en leefstijl helpt daarbij om een sterk, gebalanceerd en veerkrachtig afweersysteem op te bouwen. Door langdurig lichaamseigen blusstofjes te verbruiken kunnen toch verlaagde waarden daarvan in het lichaam ontstaan, waardoor brandjes kunnen blijven voortduren. Dan is gerichte suppletie met voedingssupplementen (zoals met PEA) de oplossing, hoe sneller hoe beter.
Dit blog gaat over medicijnverslaving, met name over hoe pijnlijk moeilijk het is om daarvan af te kicken, mijn persoonlijke verbazing over de medische adviezen om van deze verslaving af te komen, en [dus] mijn motivatie om bij te willen dragen aan het anderen helpen slagen met overwinnen van deze pijn. En wat blijkt: er is nog een wereld te winnen, en er is gelukkig ook houvast en hoop te vinden in reeds beschikbare mogelijkheden die tot nu toe - ik vind onterecht - onvoldoende benut zijn gebleven.
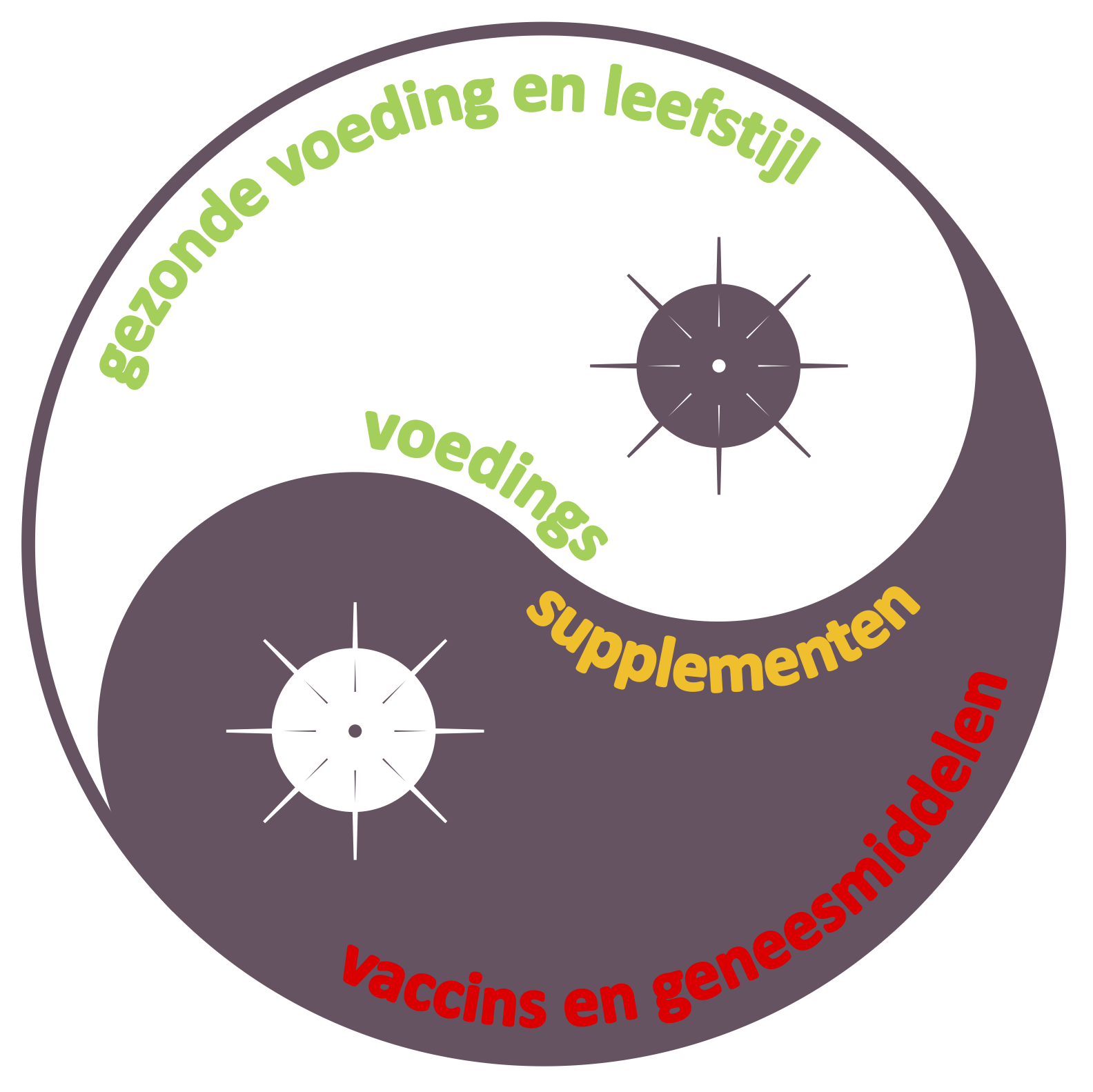
- 23 mei 2020 - Gerichte ondersteuning van gezonde voeding en leefstijl.
Zo lang het virus onder ons is zal COVID-19 de maatschappij in de greep houden, en daarbij angst en verdeeldheid blijven zaaien. Hoe lang we in die situatie zullen blijven zal in grote mate afhangen van de wereldwijde beschikbaarheid van een nieuw te ontwikkelen, veilig en effectief vaccin en/of therapie tegen het virus en de gevolgen ervan op het lichaam. Er zijn inmiddels een heel aantal vaccin-kandidaten in de race, en er worden veel nieuwe therapieën getest (I). Ook is er weer meer aandacht voor het versterken van de weerstand van ons lichaam tegen infecties en ziektes, met name door middel van gezonde voeding en leefstijlinterventies (II). De rol van voedingssupplementen om mee te strijden tegen COVID-19 komt hier nu ook aan de orde (III).
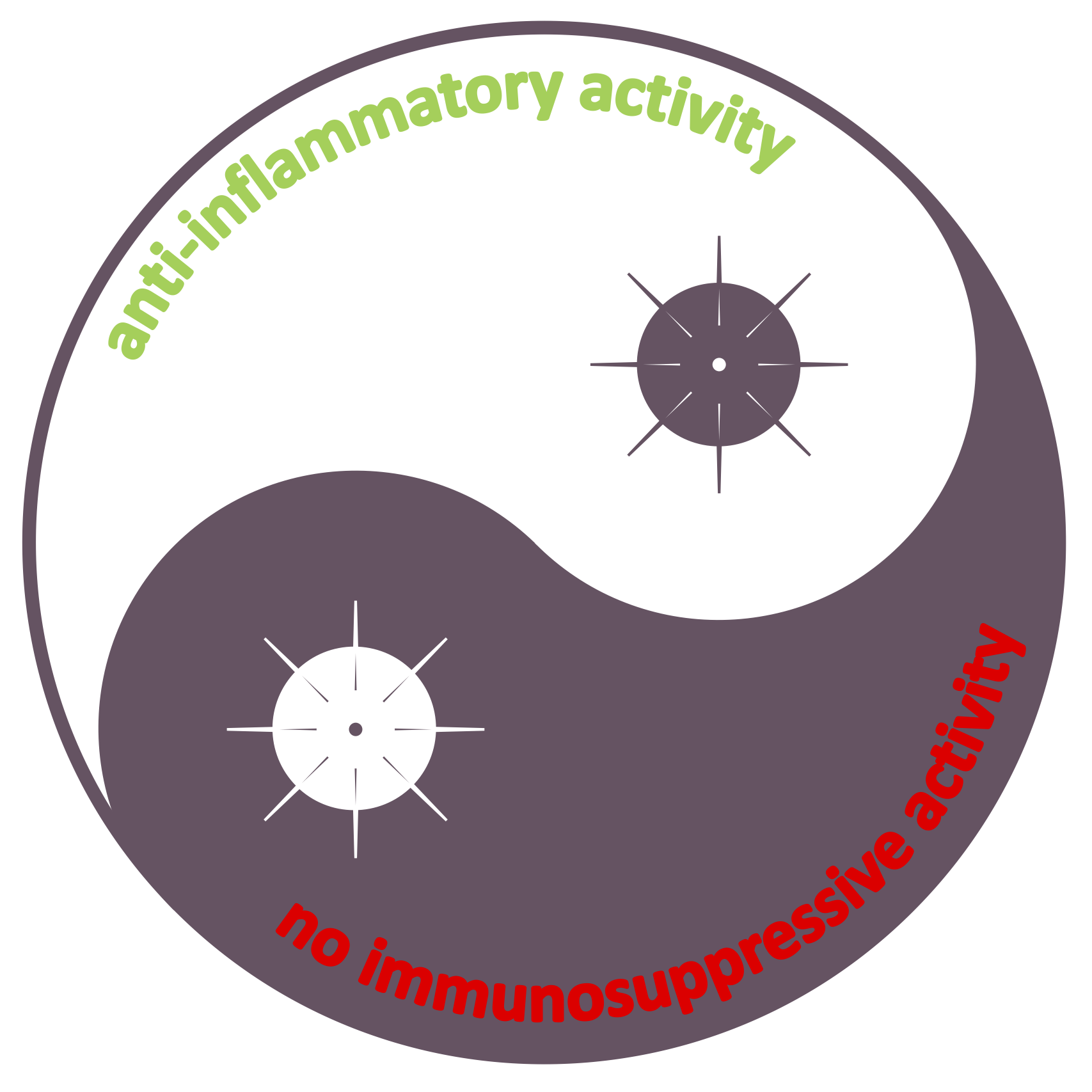
- May 12, 2020 - Soothing vascular inflammation whilst eradicating the virus.
As long as the COVID-19 disease is amongst us, and as long as there is no safe and effective vaccine, or affordable and widely applicable anti-viral drug available to all people around the globe, our world will not return to as it was before. Meanwhile the virus will continue to cause (hyper)inflammatory responses in the blood, the vasculature and thereby have major impact on the functioning of all major organs, foremost in lungs, heart, skin and brain. These responses were so far found to be most detrimental to people of older age and with underlying vascular pathologies. Yet, as long as this pandemic continues, all of us at least stand at risk of developing the perhaps milder and more manageable vascular inflammatory and thrombotic conditions, albeit with sometimes severe complications as a result (like cerebrovascular accidents / stroke).

Na jaren van zorgvuldig opbouwen is de dag gekomen om Eyesiu (lees “I see you”, oftewel ”ik zie je”; wat betekent “ik heb respect voor je”) open te stellen voor iedereen om een aandeel in het succes te kunnen nemen. Iedereen is welkom die geloof heeft in de missie en visie van de onderneming, en die vertrouwen heeft in de kunde en ervaring van de betrokken professionals. Zo help je om betaalbare innovatieve producten sneller beschikbaar te maken voor consumenten en patiënten die noodzaak of behoefte hebben aan gezondere hersenen, ogen, huid en longen.
Kom erbij en doe mee via https://www.eyevestor.com/members/eyeventures/NL_YCU
Is het lastig om pillen, capsules of vloeistoffen door te slikken? Zorgt moeite met slikken voor sociale en gezondheidsproblemen, en voor het uitstellen, weigeren of vergeten van het dagelijks innemen van geneesmiddelen of belangrijke vitamines en voedingssupplementen welke kunnen helpen het lichaam fit, sterk, pijnvrij en veerkrachtig te houden? Is meer controle gewenst over de hoeveelheid of portie van een pil of capsule die gebruikt wordt? En wil je wél snelle afgifte en opname bereiken? Dan is een toediening van de vitamines en voedingssupplementen met behulp van een handig spray flesje de ideale oplossing.
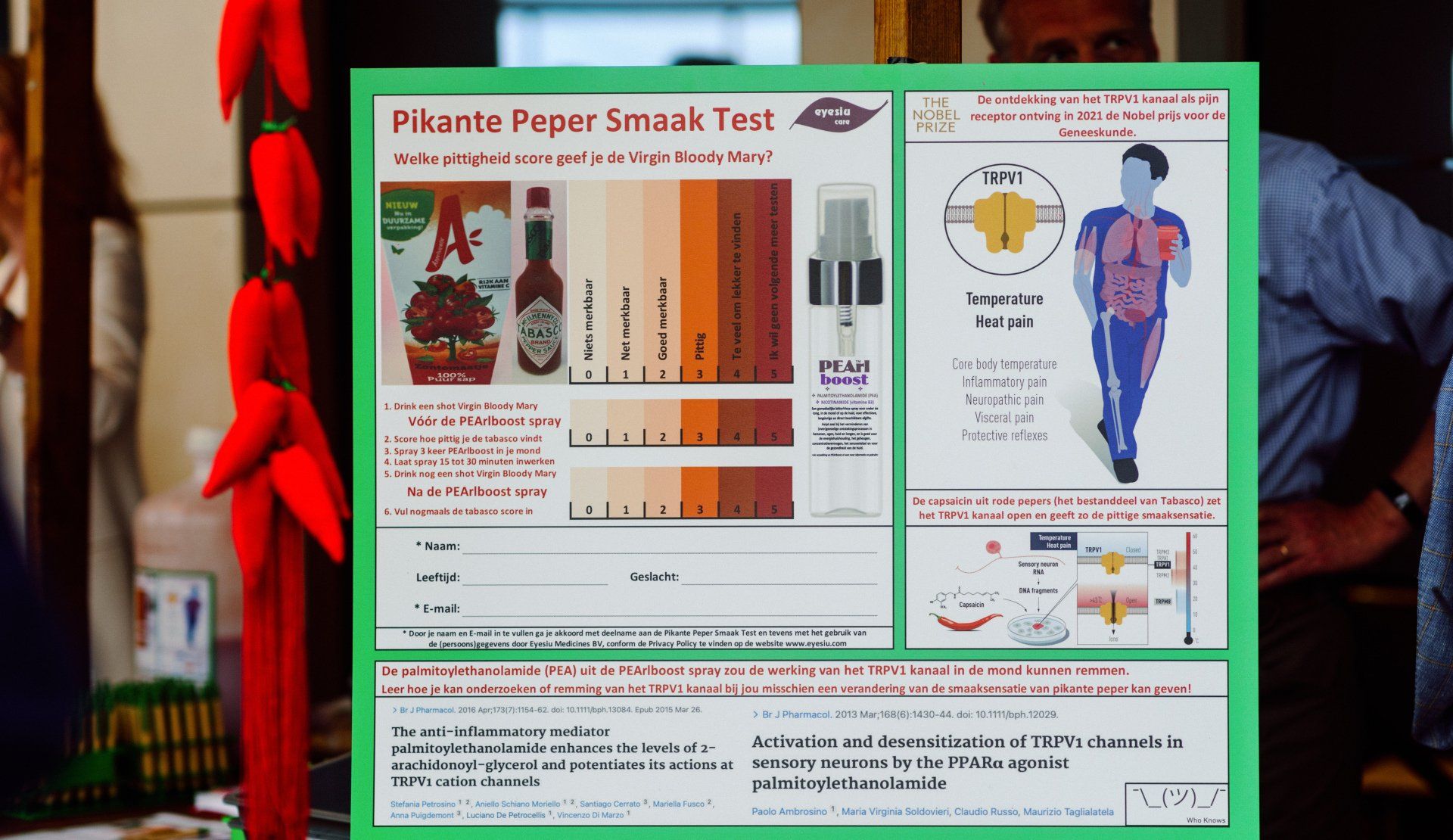
Leiden, 17 september 2022 - Tijdens de Leidse Nacht van Ontdekkingen kan je bij Eyesiu de Pikante Peper Smaakt Test komen doen. Zo ga je ontdekken welke pittigheid score jij geeft aan een shotje Virgin Bloody Mary (tomatensap met Tabasco). Hierdoor ontdek je wat activatie van het TRPV1 kanaal door de capsaïcine uit rode pepers bij jou in je mond doet op een schaal van 0 (“niets merkbaar”) tot 5 (“ik wil geen volgende meer testen”). En er zijn testflesjes van Eyesiu’s mondspray met het voedingssupplement PEArlboost++, waarvan we weten dat het ingrediënt palmitoylethanolamide (PEA) de gevoeligheid van het TRPV1 kanaal kan beïnvloeden. Dit mag je zelf testen. Zo leer je ter plekke hoe je kan onderzoeken of remming van het TRPV1 kanaal bij jou misschien een verandering van de smaaksensatie van pikante peper kan geven! Who Knows!

Op het Leids Goeds Festival van 11 mei 2022, kan je bij Eyesiu de Pikante Peper Smaak Test komen doen, en zo uitvinden welke pittigheid score jij aan een Virgin Bloody Mary geeft. Vervolgens mag je dan testen wat jouw gratis mondspray TESTFLESJE met het voeddingssupplement PEArlboost bij jou doet op je Pikante Peper Smaak Sensatie.
Het Leidsch Dagblad heeft een serie verhalen over het Leiden Bio Science Park, mede mogelijk gemaakt door het Leids Mediafonds. Het Bio Science Park is de economische motor van Leiden, de kurk waar de stad op drijft. Honderden bedrijven werken er aan de wereld van morgen. Maar wat doen ze precies? De komende maanden duikt het Leidsch Dagblad in de wereld van de biotech in de regio. Deze keer: Eyesiu, een bedrijf dat een parelmoerspray op basis van lipide microdeeltjes op de markt brengt die ontstekingsremmend werkt. En de ontstekingsremmers in de lipide nanodeeltjes zijn in ontwikkeling als geneesmiddel. Het stuk is ook te vinden op: https://www.leidschdagblad.nl/cnt/dmf20220204_24742831. Journalist & ondernemer Martijn de Meulder interviewde oprichter en directeur van Eyesiu, Pieter J. Gaillard, over zijn professionele leven, en wat inmiddels zijn levenswerk is geworden: het passeren van de bloed-hersenbarrière.


